I am not a lawyer. None of this should be interpreted as an informed legal opinion. I’m not even moderately informed about patent laws surrounding pharmaceuticals.
This is also preliminary. The post is actually going to be so short that this probably deserves to be “chat”. But most people miss those, this is too important, and I owe you people.
If you read Moberg’s quarterly & annual reports, the following is basically all they say.
“The most important markets for MOB-015 are expected to be the U.S., EU, Japan, Canada and China, all with approved patent protection through to 2032. In addition to granted patents, the company also has ongoing patent applications which, if approved, could provide significantly longer patent protection.”
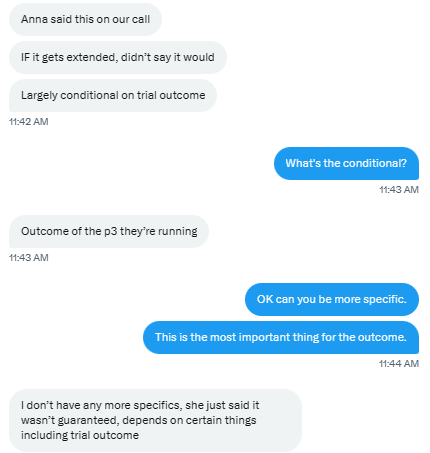
What does significantly longer mean? And what is meant by ongoing patent application?
That’s the first hint. It doesn’t really sound like they intend to pursue patent extensions to extend patent protection. It sounds like they’re trying to cover something else about the product, and it sounds like the 2nd phase 3 readout could somehow be involved.