There’s always been something weird about how these trials were constructed.
If you look at the trial construction closely, it is liable to being influenced by something other than just very large concentrations of terbinafine being measured in the nail plate and nail bed from prior use. They weren’t just going from 48 weeks to 8 weeks on a whim, they were testing their own drug and learning about one thing in particular: the whitening.
The claims around the maintenance phase being sufficient are within a dosing range, so you need to wrinkle your pigly-wigglies a bit to understand what they’re trying to do. The argument is basically they’ve made a novel delivery mechanism for terbinafine that relates to the aesthetic change the nail undergoes.
This is what they want to patent.
Moberg’s interest in their IP likely has everything to do with the construction of how aggressive this trial was. You can read the patent here.
8 weeks of dosing has probably elucidated the clinical importance of this process in clinical trial, but only if mycological cure rates are now low. This “whitening” and the near universal tipping point of the nail experiencing it, isn’t at 8 weeks of daily use. It happens gradually over time and becomes most notable for most users at week 12, and Moberg knows this.
Isn’t that interesting?
If granted this would obviously extend the lifespan of the asset.
I will be able to know once I’ve tested the product on enough non-diseased nails, but I believe the products “whitening” has some important interplay with the rate of the ongoing cell death of the fungus.
I don’t expect my nails will be turning the same color as a nail fungus patient for this reason either. This makes me smile because it kind of makes me think the whitening could, in part, be a smooth homogenous blend of eukaryotic material staining the reflective surface of the nail plate. Like blood on cloth.
This is important as this patent was Moberg’s plan to protect their IP. Nothing explicitly required this readout to be particularly good to achieve this end. Extending the lifespan of their asset overwhelms the financial incentives to positively effect the label in the short run.
From blinded to unblinded - learning more
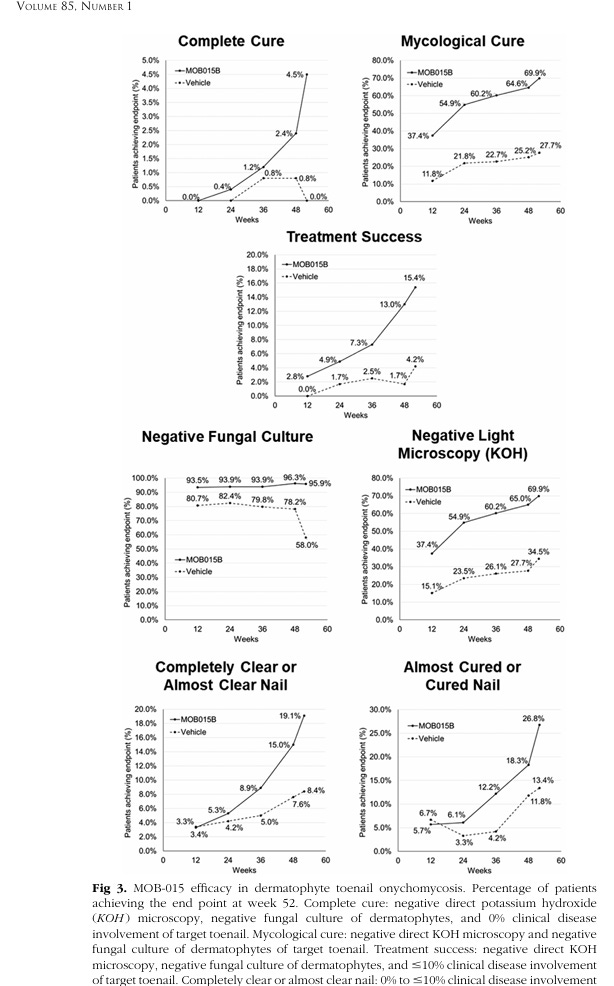
When Moberg unblinds the data of this P3 trial, I expect we’ll be seeing a somewhat similar starting point to the last. Below is the prior trial data for the curves of interest.
The washout period revealed these curves to be exponentiating, so it’s a natural conclusion to draw that they needed to just reduce the dosing. But they pulled back a lot. Now what 8 weeks looks like, more closely, is the delineation of the boundary of this hydrated state and its relationship to product efficacy.
Below you will see the products use, some of whom modified their dosing. All are still under 9 months of use. Earliest consumer access to the product was in January, but launch happened in February and most consumers will have only been using the product since April.
There are more photos than the ones below, however, the ones below have also been willfully shared publicly. It’s most clear what I’m talking about with the last set of photos and the 2nd last photo.










I have emailed Anna asking her to provide some colour regarding the trial design strategy. What you have outlined above is the only logical explanation to me. The only alternative I can come up with is that they used previous trial data to come up with a dodgy statistical model that gave them false confidence that an 8-week loading phase would yield competitive cure rates, but it still sounds too aggressive for this management team.
If they do have a strategy here it is very frustrating that they haven't communicated it previously.
It sounds like the Friday 13th PR was because of an error, in which case Anna is certainly walking on regulatory eggshells to maintain the trial integrity. I am not sure that it would be appropriate for her to communicate contingency plans now because it might convey a certainty of trial failure (as opposed to just a 'lowered expectation'). Although Occam's razor says this is just another display of woeful management.
Do we have an idea of when we will know if this plan to extend protection of their IP was successful? If it works, it would be a good outcome. I know ow nothing about patent applications and approval processes. Are we talking international protection or only in the states?